Page name
GLP-1 Drug Demand and Its Impact on Pharmaceutical Logistics Hiring
The clamor for GLP-1 medicines such as Ozempic®, Wegovy® and Mounjaro® has dominated most of the conversations I’ve had in the last few months.
What we’re seeing in the market is a steady increase in temperature-controlled shipments moving through global distribution networks. Pharmaceutical manufacturers are expanding production capacity and logistics providers are adjusting their infrastructure, warehouse capacity and transport networks to support that demand.

How Do GLP-1 Medicines Affect Pharma Logistics?
Pharmaceutical logistics involves transporting medicines under strict regulatory and temperature-controlled conditions. GLP-1 drugs typically need to be kept between 2°C and 8°C, which means cold chain integrity must be maintained across international transport, warehousing and distribution.
Temperature-controlled packaging providers such as Peli BioThermal, Emball’iso and Envirotainer support this process, alongside specialist courier networks including Marken and World Courier that handle time-sensitive pharmaceutical shipments.
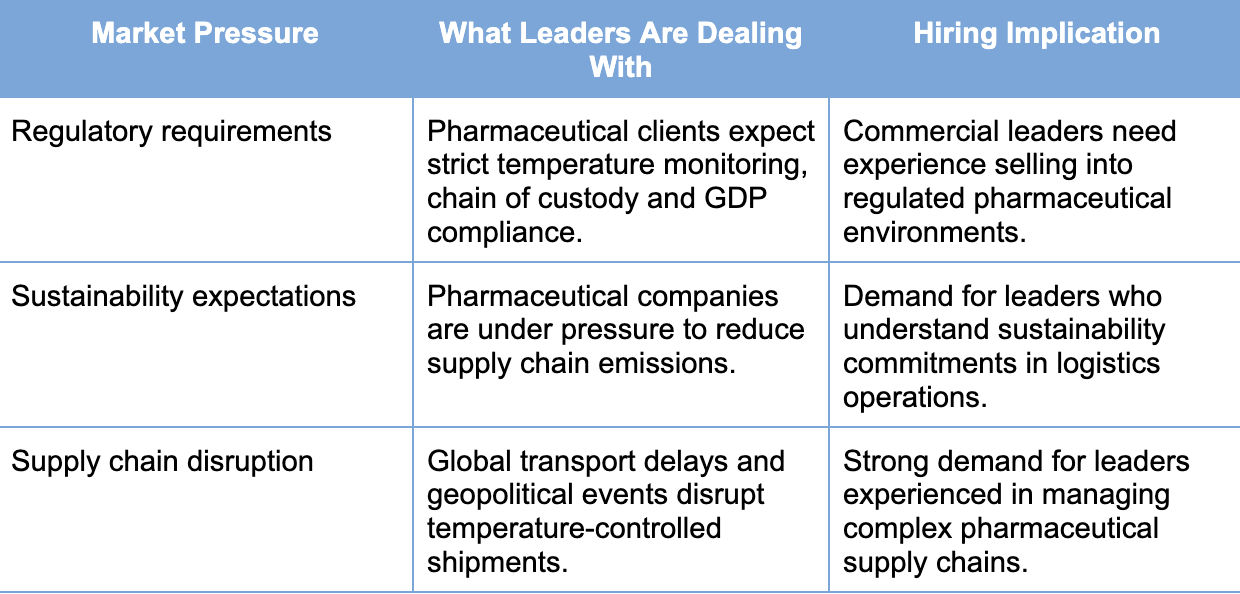
Sales leaders in pharmaceutical logistics are balancing three pressures at once: GLP-1 demand, regulatory complexity and rising expectations around sustainability.
What Does This Mean for Cold Chain Logistics?
Pharmaceutical manufacturers are expanding production capacity to meet demand for GLP-1 medicines.
Novo Nordisk and Eli Lilly have both announced significant investment into manufacturing facilities and research infrastructure to support demand for these treatments.
Companies are investing in packaging technology, monitoring systems and pharmaceutical warehousing to support that growth. But in conversations with operations leaders, the bigger constraint often comes down to hiring people who understand pharmaceutical logistics and can manage that scale while maintaining compliance.
What These Market Pressures Mean for Hiring
As demand for GLP-1 medicines grows, pharmaceutical logistics companies are scaling operations to handle higher volumes of temperature-controlled shipments. That means expanding cold-chain capacity, managing transport disruption and meeting rising sustainability expectations from pharmaceutical clients.
These pressures also affect the commercial side of the business. Sales and operations leaders need to understand pharmaceutical regulation, how pharma companies buy logistics services, and the risks involved in complex supply chains.
As a result, these industry pressures are shaping how pharmaceutical logistics companies prioritise hiring. Many candidates come from general freight backgrounds but have limited experience working within regulated pharmaceutical environments.
Market Pressures and Their Hiring Impact
Looking Ahead
As pharmaceutical manufacturers continue expanding production of GLP-1 medicines and other biologic therapies, logistics providers will need to scale temperature-controlled distribution while maintaining strict regulatory standards.
For pharmaceutical logistics companies, that demand creates opportunity.
If you work in pharmaceutical logistics or temperature-controlled supply chains, feel free to connect with Adam Dowson or the Spencer Riley team to discuss what you’re seeing in the market.
Contact us today
Spencer Riley’s team of highly dedicated, specialist consultants’ pride themselves on gaining a full understanding of our client’s business,







