Page name
Is GLP-1 Demand Changing the Clinical Trials Market?

GLP-1 drug demand is changing how pharmaceutical companies invest, reducing clinical trial activity in some areas and increasing pressure on patient recruitment and delivery.
While much of the attention has focused on manufacturing and supply, early changes are now visible in the development cycle. Pharmaceutical companies are reallocating R&D capital, and clinical research activity is shifting as a result.
What We’re Seeing in the Market
Across conversations with senior leaders in pharmaceutical services and clinical research, a consistent pattern is emerging.
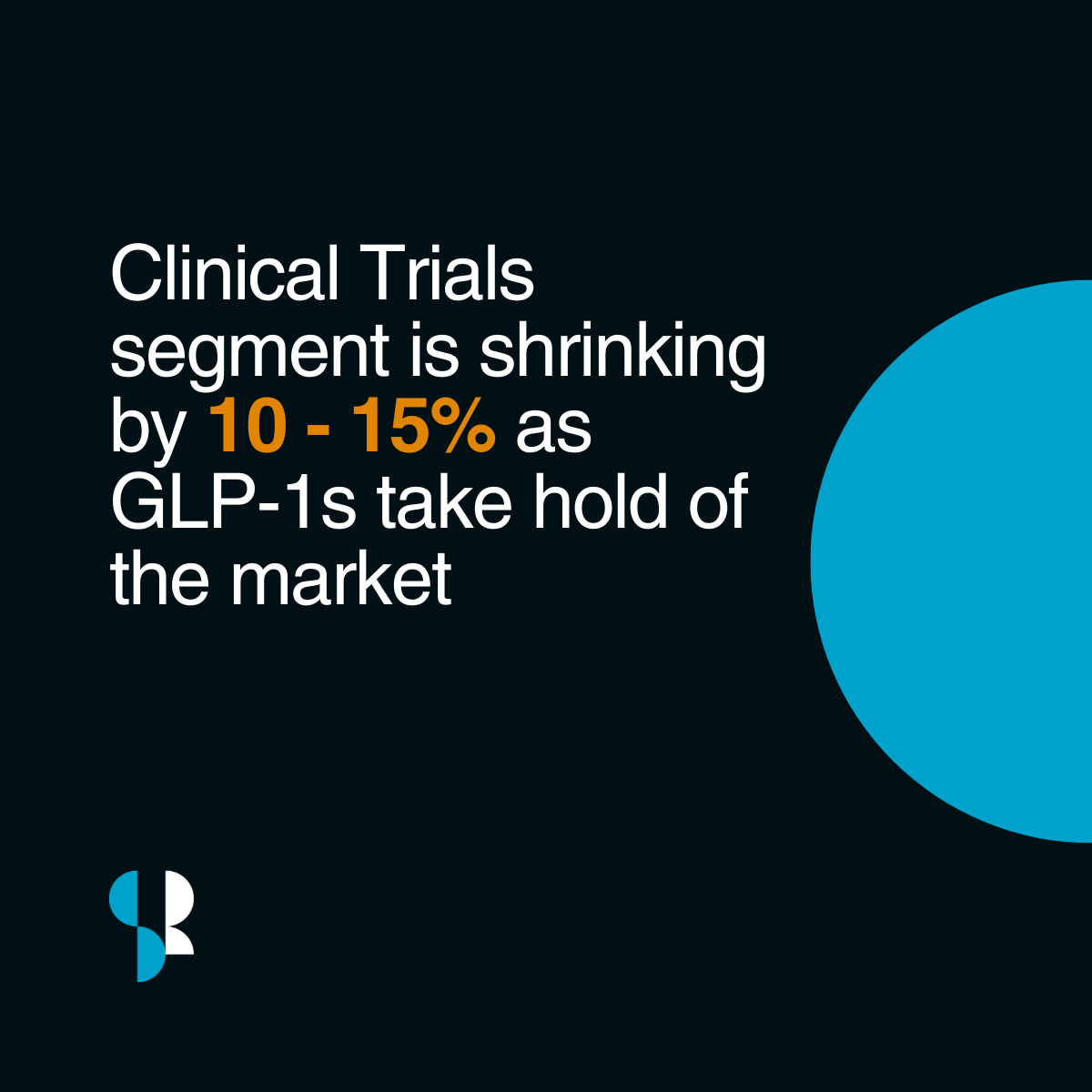
In some areas of clinical research, activity is slowing. Estimates suggest parts of the clinical trials market could contract by around 10–15% as investment shifts towards established drug classes like GLP-1s.
Three factors driving change:
1. Investment is concentrating around GLP-1 drugs
Pharmaceutical companies are prioritising established, high-demand treatments, reducing funding available for other pipelines.
2. GLP-1 drugs are quicker and cheaper to develop
Compared to entirely new pipelines, these treatments require less time and lower investment, changing how pharmaceutical companies allocate R&D budgets.
3. Patient recruitment is becoming more difficult
Around 80% of trials experience delays, with enrolment and retention cited as primary causes. Post-COVID, participation has become more challenging, increasing costs and extending timelines.
I explored the supply-side impact of this trend in our earlier article on GLP-1 demand and pharmaceutical logistics hiring.
What This Means for Clinical Service Companies
For CROs and clinical service organisations, the impact is uneven.
Some teams are seeing strong demand linked to high-growth therapeutic areas. Others are operating in a more competitive environment, with increased competition for available studies and greater pressure on pricing and timelines.
Recent industry commentary, including reports of restructuring across parts of the CRO market, suggests demand is shifting rather than declining overall.
In conversations with leaders, the focus is shifting toward:
- Securing work in therapeutic areas with sustained investment
- Improving efficiency in trial delivery
- Managing tighter margins and longer sales cycles
This reflects a market that is not shrinking overall, but becoming more selective.
What This Means for Hiring
As investment shifts towards GLP-1 and trial activity becomes more selective, hiring strategies need to adapt.
- Focusing hiring in therapy areas where investment is increasing
- Prioritising candidates who can manage complex trials and tight timelines
- Strengthening clinical operations teams to handle ongoing patient recruitment challenges
For leaders, the priority is hiring people who can deliver trials efficiently and understand the commercial pressures behind them.
If you're hiring within clinical research, CROs, or trial operations, connect with Adam Dowson or the Spencer Riley team to build the teams needed to support this change and deliver long-term growth.
Contact us today
Spencer Riley’s team of highly dedicated, specialist consultants’ pride themselves on gaining a full understanding of our client’s business,